A new solution to get athletes with shin splints back on track faster
Ask any runner about injuries, and there’s a good chance they’ll mention shin splints – a painful condition that strikes in up to a third of athletes each year. If left untreated, shin splints – also called medial tibial stress syndrome – can cause stress fractures.
Despite any number of devices on the market that purport to get an athlete back on track quickly, the only treatment backed by evidence is rest: weeks, sometimes months, sidelined from training. This forced break can end an athlete’s season.

Photo supplied: Ben Lindsay
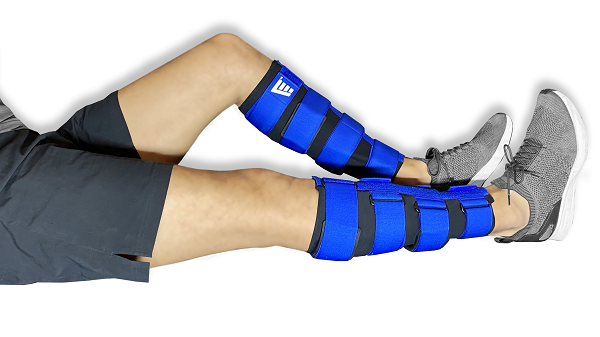
So in 2015, to get people afflicted with shin splints up and running again sooner, three then-undergraduate students got together: national finalist runner and now doctor Dr Will McNamara; designer and national finalist gymnast Rosa Miller; and engineer and international finalist swimmer Ben Lindsay. They started developing a device that combines soft-tissue therapy with bone-remodelling treatments and called it the Solushin.
“Our main goal was to reduce the rest required and to keep athletes training hard the whole year round,” Lindsay says. And a randomised, double-blind, controlled trial at Sydney Sports Medicine Centre found the Solushin did just that; it got athletes back to pain-free training within five weeks. But even though they had an evidence-backed device on their hands, the inventors realised that they needed help getting it to market.
So Lindsay and Miller completed the Medical Device Commercialisation Training Program (MDCTP) in 2019 – a 12-week business-building courses and workshop series focussing on clinical trials, regulatory strategy and intellectual property. It was an experience that completely changed their product and business approach.
“We came in with the very naïve view that we already knew the market, but that was quickly broken through the interview part of the course where we had to go and talk to people,” Miller says. “That was one of the most valuable parts of the program – breaking those assumptions and teaching us that we didn’t know everything.”
Shin splints appear more often in amateur runners, so Miller and Lindsay naturally thought the best way to reach that cohort was to sell their product relatively cheaply. But that assumption was flipped after speaking with amateur runners, who told Miller and Lindsay that price wasn’t their main consideration. Instead, they favoured products used and endorsed by professionals.
“Then we found out through a pricing analysis taught in the program that elite runners, triathletes and physios expected a higher-quality product,” Lindsay says.
So by raising the Solushin’s retail price, the inventors had the financial freedom to spend more on its manufacture, and partner with NSW-based suppliers, to produce a premium product – something that they couldn’t have achieved if they stuck to a lower price.
Even though the MDCTP focused on medical devices, Lindsay adds that the skillset he learnt from it “could apply to just about any product you wanted to build”.
Miller agrees: “You can adapt it to any situation, really, and I now feel more educated in understanding what a good versus a bad product is.”
The coming months are set to be a busy time for the trio. Their first manufacturing run of the Solushin kicked off in January and they recently won a Minimum Viable Product grant from the NSW Government. “Everything I put in that grant application was what I learnt in the program,” Lindsay says.
“I think we got halfway through the program when we were like, ‘Oh, this is actually going to work’,” Miller says. “That was a real lightbulb moment for us: we were putting all the work together and restructuring our business plan to be a more streamlined process of getting the product to market.”
By Bel Smith
Updated 6 months ago